 
CO 2 has one carbon atom and two oxygen atoms.Oxygen (O) has an atomic mass of about 16.00 amu.Carbon (C) has an atomic mass of about 12.01 amu.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound.Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound. 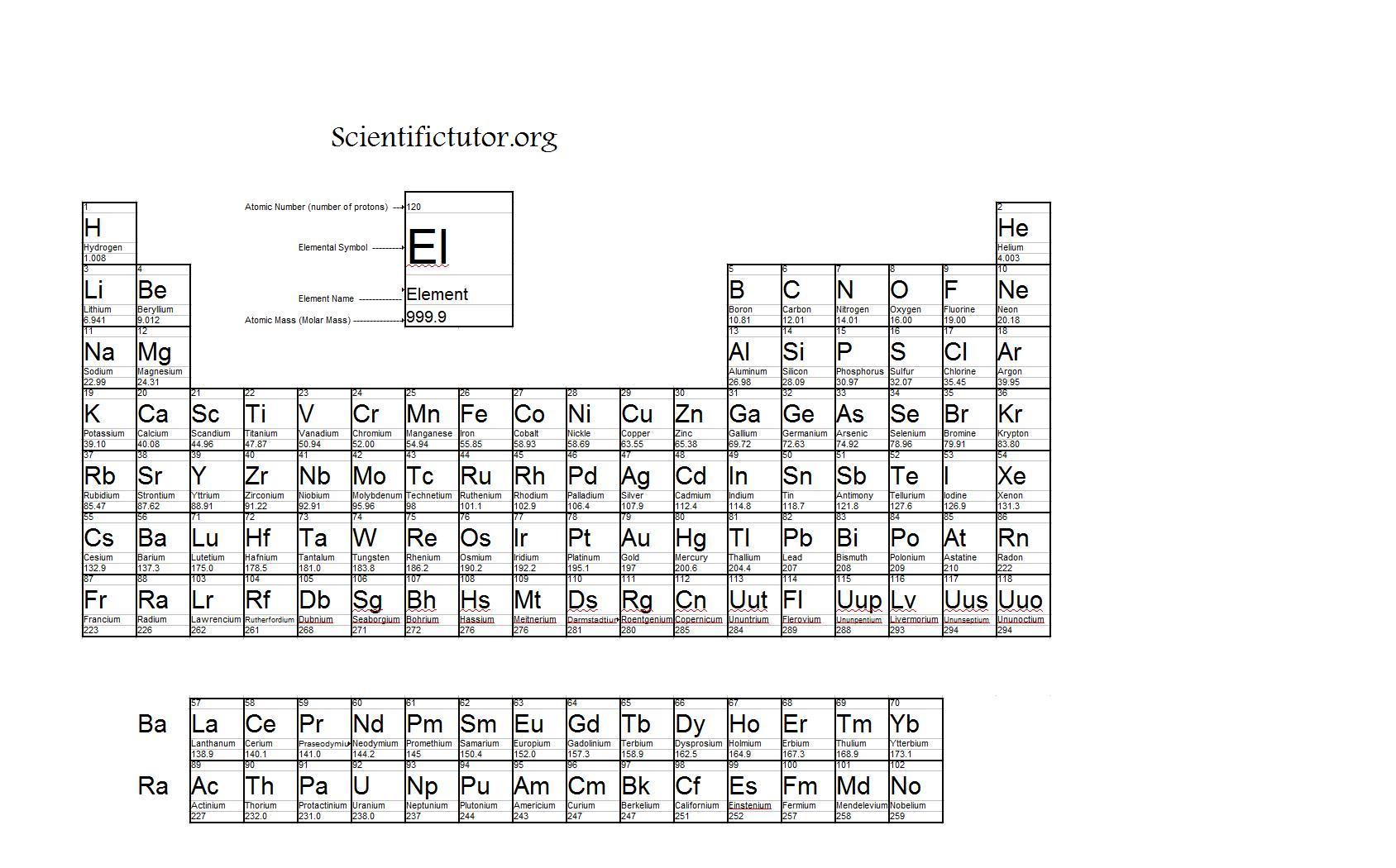
Its by far the most abundant element in its group in the periodic table and yet it is the last member of its family to be discovered. The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). A measure of how much energy is needed to break all of the bonds of the same type in one mole of gaseous molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
